Transcranial Magnetic Stimulation, commonly called TMS, is one of the most significant developments in modern psychiatric treatment and interventional psychiatry. This guide is designed to serve as a long-term, patient-facing reference page grounded in evidence and written in plain English.
TMS has been studied for decades and is now widely used in outpatient psychiatric settings. Below, you will find a structured, overview of what TMS is, how it works, what it treats, how effective it is, and what you should consider before starting treatment.
Image Source: www.magventure.com
What Is TMS (Transcranial Magnetic Stimulation)?
TMS stands for Transcranial Magnetic Stimulation. It is a noninvasive medical treatment that uses magnetic pulses to stimulate specific areas of the brain involved in mood regulation and other cognitive functions.
TMS does not involve surgery. It does not require anesthesia. It does not involve systemic medication circulating through the bloodstream.
The treatment was first cleared by the U.S. Food and Drug Administration (FDA) in 2008 for adults with major depressive disorder who had not responded adequately to antidepressant medication. Since then, additional indications have been cleared, and research has expanded significantly.
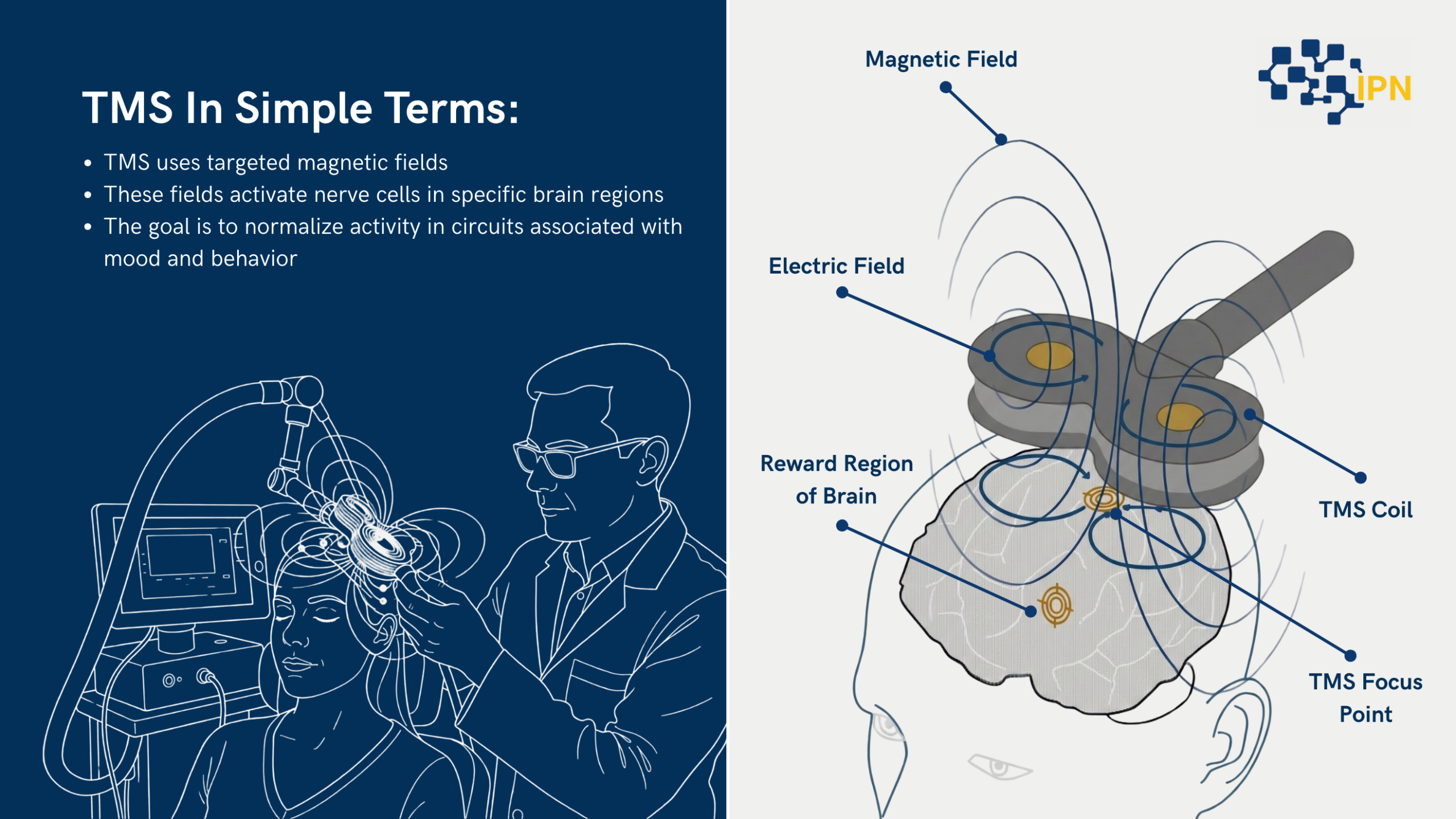
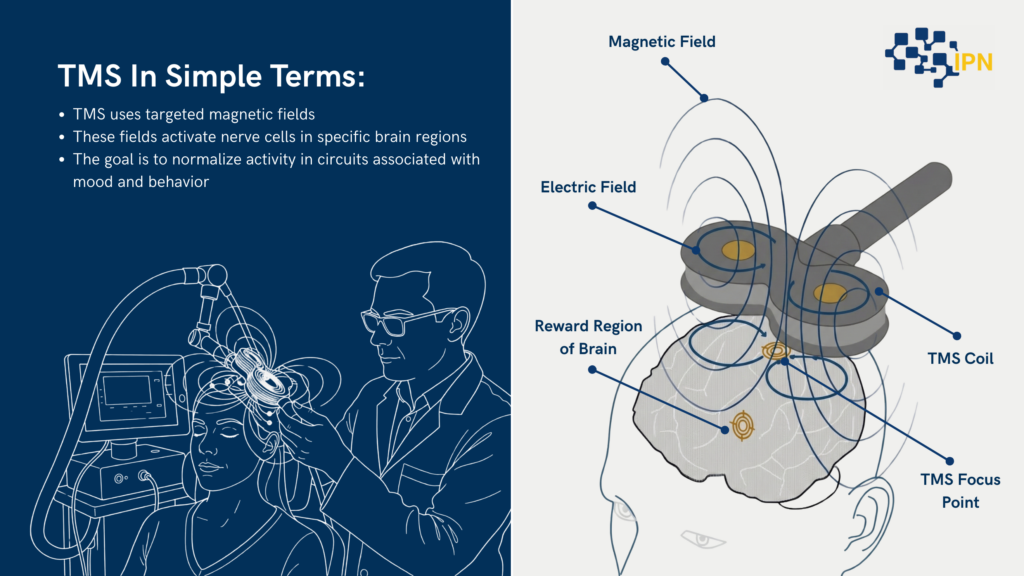
In simple terms:
- TMS uses targeted magnetic fields
- These fields activate nerve cells in specific brain regions
- The goal is to normalize activity in circuits associated with mood and behavior
Unlike medications, which affect the entire brain and body, TMS targets a defined area of the cortex, most commonly the left dorsolateral prefrontal cortex, a region consistently implicated in depression.
Today, TMS therapy is offered in outpatient psychiatric and neurology settings across the United States and internationally.
Latest Articles
Join Our Email List
Sign up to receive the latest articles right in your inbox.
How TMS Works
To understand what TMS does, it helps to understand one basic concept about depression and other psychiatric conditions: certain brain circuits can become underactive or dysregulated.
In major depressive disorder, research using functional imaging has shown that specific frontal brain regions often show reduced activity. These areas are connected to deeper limbic structures involved in emotion regulation.
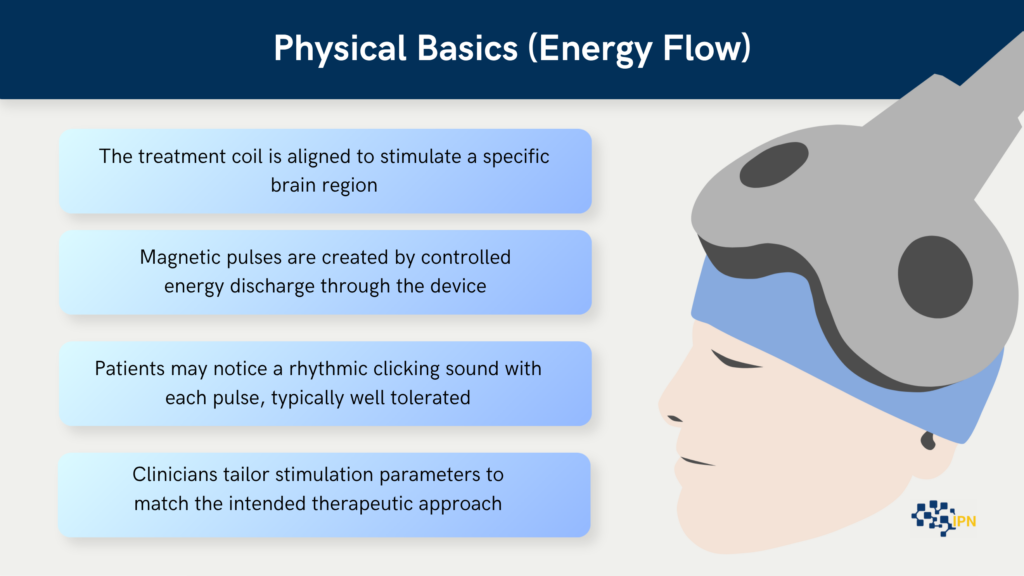
TMS works by:
- Placing a magnetic coil against the scalp.
- Delivering brief magnetic pulses.
- Generating small electrical currents in the underlying brain tissue.
- Activating neurons in targeted networks.
These magnetic pulses pass through the skull painlessly. They do not require any incision. The magnetic field induces a small electrical current in the brain tissue directly beneath the coil. This process is called electromagnetic induction.
Repeated stimulation over weeks can lead to measurable changes in brain network activity. In clinical practice, this is referred to as neuromodulation.
Over time, repeated sessions appear to help “reset” or rebalance dysfunctional circuits, especially in people whose depression has not responded to medication.
It is important to understand that TMS does not “shock” the brain. It is fundamentally different from electroconvulsive therapy. The stimulation is focal, controlled, and does not induce seizures in standard depression protocols.’
What Conditions TMS Is Used For
TMS therapy has both FDA-cleared uses and areas of active research. It is essential to distinguish between these.
FDA-cleared indications
In the United States, the FDA has cleared TMS for:
Major Depressive Disorder (MDD)
TMS for depression is the most established and widely used indication. It is typically used in adults who have not experienced adequate improvement from antidepressant medications.
Obsessive-Compulsive Disorder (OCD)
A specific TMS protocol targeting different brain regions has been cleared for adults with OCD.
Smoking Cessation
Certain TMS systems have received FDA clearance for smoking cessation support.
Migraine with aura
A single-pulse TMS device has been cleared for acute treatment of migraine with aura.
Each of these indications has specific protocols, coil placements, and stimulation parameters.
Off-label research (clearly labeled, cautious)
There is ongoing research investigating TMS in:
- Bipolar depression
- Post-traumatic stress disorder (PTSD)
- Anxiety disorders
- Chronic pain
- Substance use disorders
- Cognitive impairment
These uses are considered off-label unless specifically cleared by the FDA. That does not mean they are ineffective, but it does mean the evidence base varies and protocols are still evolving.
Patients considering TMS for off-label conditions should ask:
- What evidence supports this use?
- Is this based on randomized controlled trials?
- How does the clinic define success?
An evidence-first approach is essential.
What TMS Treatment Is Like
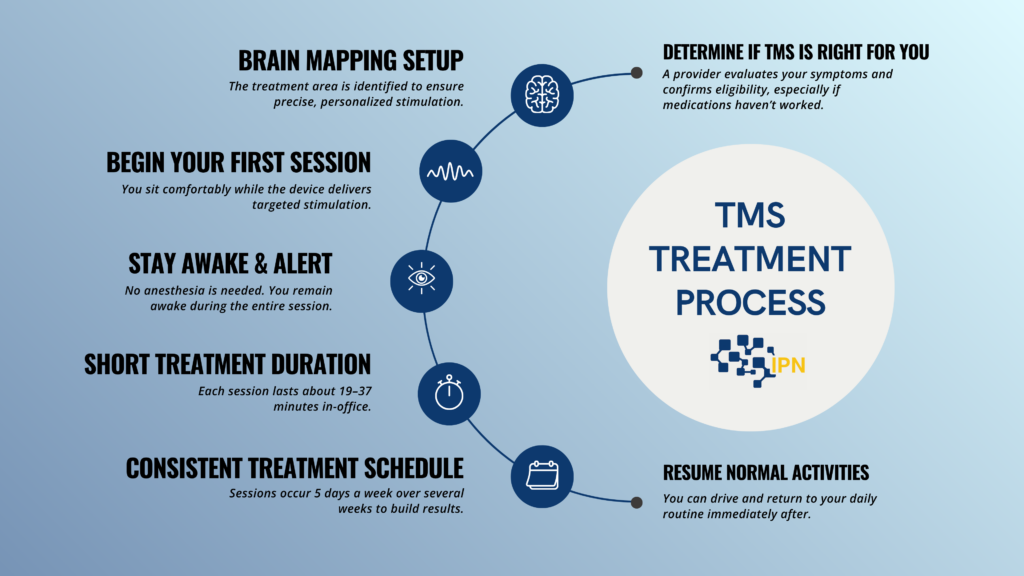
One of the most common questions is: What is TMS therapy actually like?
Before treatment begins
Patients typically undergo:
- A psychiatric evaluation
- Review of medical history
- Medication history
- Screening for contraindications
During the first session, clinicians determine the motor threshold. This is the minimum level of stimulation required to produce a small movement, usually in the hand. This helps individualize treatment intensity.
During a session
A typical TMS session involves:
- Sitting in a comfortable chair
- A magnetic coil positioned on the scalp
- Repetitive clicking sounds from the device
- A tapping sensation on the scalp
Patients remain awake and alert throughout. They can return to work or normal activities immediately after.
There is no sedation. No recovery period is required.
Treatment schedule
Standard TMS for depression usually involves:
- 5 sessions per week
- For about 4 to 6 weeks
- Sometimes followed by a taper phase
Each session lasts approximately 20 to 40 minutes, depending on the protocol.
Some newer accelerated protocols are under investigation and in limited use, but traditional daily sessions remain common.
What patients typically report
During sessions:
- Mild scalp discomfort, especially early on
- Tapping or knocking sensation
- Gradual accommodation as treatment progresses
After sessions:
- Most patients resume daily activities immediately
- No cognitive impairment
- No need for transportation assistance
Improvement is often gradual. Some people begin to notice changes in mood after 2 to 3 weeks, though responses vary.
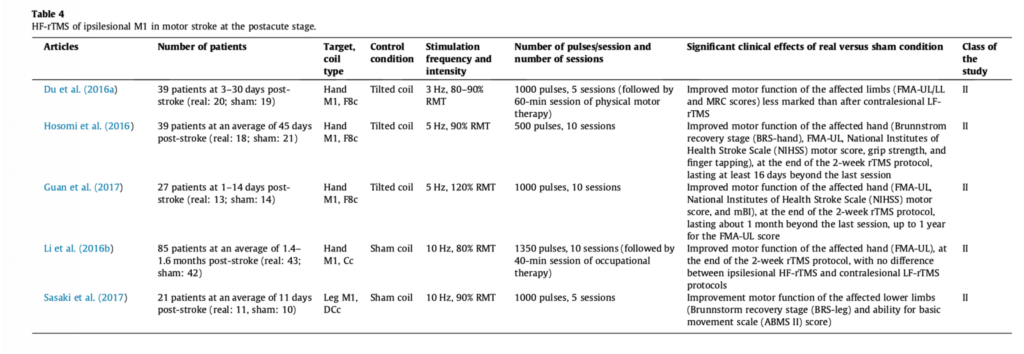
Image Source: Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS)
How Effective Is TMS?
When people ask, “Does TMS work?” they are really asking two different questions:
- What do controlled studies show?
- What happens in real-world clinical settings?
What studies show
Randomized controlled trials have demonstrated that TMS therapy can significantly reduce depressive symptoms in individuals with treatment-resistant depression.
In many large studies:
- Response rates (meaning significant symptom improvement) range from about 50 to 60 percent.
- Remission rates (meaning minimal or no symptoms) often range from about 30 to 40 percent.
These numbers vary depending on:
- Study design
- Patient population
- Severity of depression
- Number of prior medication failures
Meta-analyses have consistently shown that active TMS outperforms sham (placebo-like) stimulation in depression.
What varies in real-world settings
In everyday practice, outcomes depend on:
- Accurate diagnosis
- Appropriate patient selection
- Protocol quality
- Adherence to full treatment course
Patients with multiple prior medication failures may still respond, but overall remission rates can vary.
It is also important to understand that TMS is not guaranteed to work. Some patients do not respond. Others experience partial improvement.
For those who respond, benefits can last months to years. Some patients may benefit from maintenance or repeat courses if symptoms return.
Safety, Side Effects, and Risks
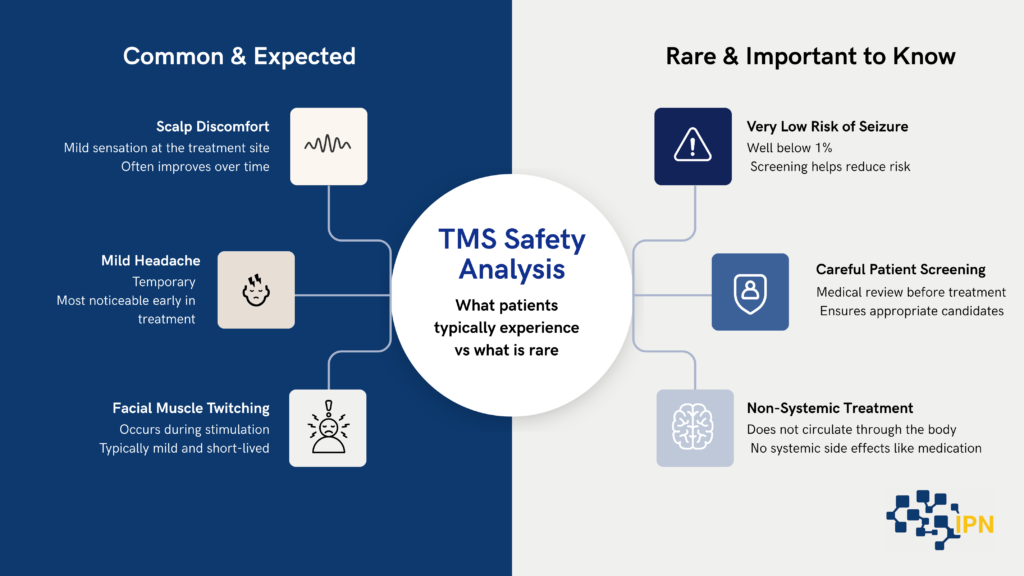
Source: Safety and recommendations for TMS Rossi Research
One of the reasons TMS therapy has gained attention is its favorable safety profile compared to many other psychiatric interventions.
Common side effects
The most common side effects include:
- Scalp discomfort at the treatment site
- Mild headache
- Facial muscle twitching during stimulation
These effects are typically:
- Mild to moderate
- Temporary
- Most noticeable early in treatment
They often diminish as sessions continue.
Less common risks
The most serious known risk of TMS is seizure. However:
- The risk is very low.
- It is estimated at well below 1 percent.
- Proper screening and adherence to guidelines significantly reduce risk.
TMS does not cause:
- Memory loss
- Systemic side effects like weight gain or sexual dysfunction
- Cognitive impairment
Unlike medications, TMS does not circulate through the bloodstream, so it does not affect organs such as the liver or kidneys.
Who should not receive TMS
TMS is generally not recommended for individuals with:
- Certain implanted metallic devices in or near the head
- Deep brain stimulators
- Cochlear implants
- Some types of aneurysm clips
A thorough medical review is necessary before starting.
Who May or May Not Be a Good Candidate
TMS is most commonly considered for:
- Adults with major depressive disorder
- Individuals who have not responded adequately to at least one antidepressant
- People who cannot tolerate medication side effects
- Individuals seeking a non-systemic option
TMS may be especially appealing to patients who:
- Want to avoid medication changes
- Have experienced medication side effects
- Prefer a noninvasive outpatient treatment
However, TMS may not be ideal for:
- Individuals with unstable medical conditions
- Those unable to attend frequent sessions
- Patients with certain neurological risks
A comprehensive psychiatric evaluation is essential before making a decision.
TMS Compared to Other Treatments
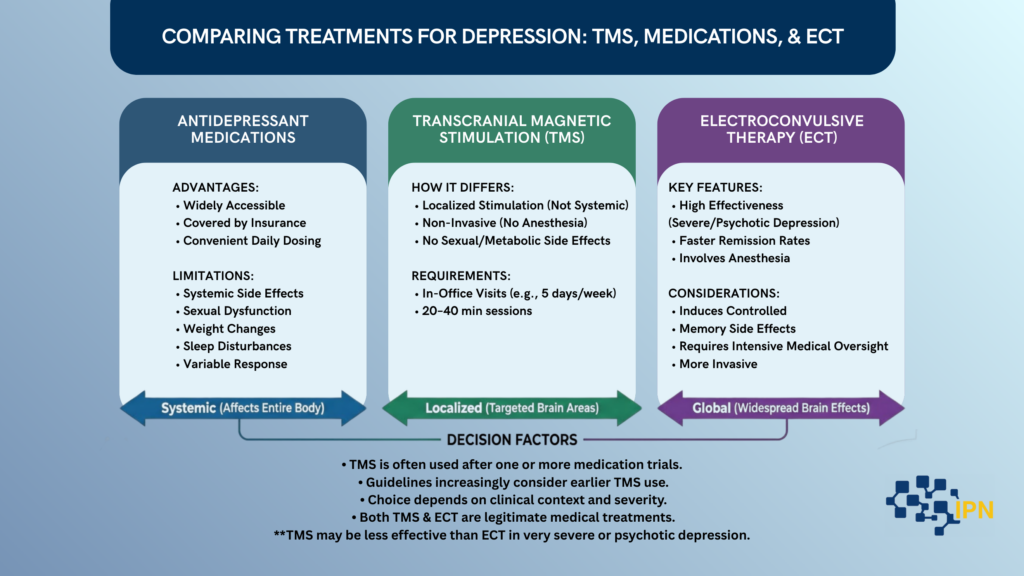
Understanding how TMS compares to other treatments can help patients make informed choices.
Medications
Antidepressants remain first-line treatment for many individuals with depression.
Advantages of medication:
- Widely accessible
- Covered by most insurance plans
- Convenient daily dosing
Limitations:
- Systemic side effects
- Sexual dysfunction
- Weight changes
- Sleep disturbances
- Variable response
TMS differs in that it:
- Is localized rather than systemic
- Does not typically cause sexual or metabolic side effects
- Requires in-office visits
TMS is often used after one or more medication trials, though some guidelines increasingly consider earlier use.
ECT (high-level, neutral)
Electroconvulsive therapy (ECT) is another neuromodulation treatment used for severe depression.
ECT:
- Involves anesthesia
- Intentionally induces a controlled seizure
- Is highly effective, especially in severe or psychotic depression
ECT may produce faster and higher remission rates in severe cases.
However, ECT can be associated with:
- Memory side effects
- Need for anesthesia
- More intensive medical oversight
TMS:
- Does not require anesthesia
- Does not induce seizures in standard protocols
- Has a milder side effect profile
- May be less effective than ECT in very severe or psychotic depression
Both are legitimate medical treatments. The right choice depends on clinical context.
What TMS Does Not Mean (Limits and Misconceptions)
As interest in TMS therapy has grown, so have misunderstandings about what it is and what it is not. Clarifying these distinctions helps patients form realistic expectations.
TMS is not brain surgery. It is a noninvasive outpatient treatment. The magnetic pulses pass through the scalp and skull without any incision, implant, or alteration of brain tissue. Patients remain awake and alert throughout the procedure.
TMS is also not electroconvulsive therapy. Unlike ECT, standard TMS protocols do not induce seizures and do not require anesthesia. The stimulation used in TMS is focal and controlled, targeting specific cortical regions rather than producing a generalized seizure.
It is important to understand that TMS is not an immediate solution. While some individuals report early improvement, most experience gradual symptom change over several weeks of consistent treatment. Response patterns vary, and full benefit often emerges toward the latter half of a treatment course.
TMS does not replace psychotherapy. The two approaches serve different but complementary roles. TMS targets neural circuitry associated with mood regulation, while psychotherapy addresses thought patterns, behaviors, coping strategies, and life context. Many patients benefit from combining both.
Finally, TMS is not guaranteed to work. Some individuals experience significant improvement or remission, others experience partial relief, and some do not respond. As with all medical treatments, outcomes vary. A careful evaluation and realistic expectations are essential before beginning therapy.
Common Questions About TMS (FAQ)
What is TMS in simple terms?
TMS is a noninvasive treatment that uses magnetic pulses to stimulate specific areas of the brain involved in mood regulation.
Is TMS therapy painful?
Most patients describe it as a tapping sensation on the scalp. Discomfort is usually mild and temporary.
How long does TMS take to work?
Some people notice improvement after 2 to 3 weeks. Others require the full course before changes become clear.
Is TMS covered by insurance?
Many insurance plans cover TMS for depression when certain criteria are met, such as prior medication trials.
Can TMS be repeated?
Yes. Some patients undergo repeat courses if symptoms return.
Does TMS cause memory loss?
Unlike ECT, TMS has not been shown to cause memory loss in standard depression protocols.
What happens if TMS does not work?
If there is no improvement, clinicians may reassess diagnosis, consider alternative protocols, or explore other treatments.
How do I get TMS treatment? Do I need a referral?
Access to TMS therapy depends on the clinic, your insurance plan, and state regulations. In most cases, patients begin with a comprehensive psychiatric evaluation to determine whether TMS is clinically appropriate. Some clinics require a referral from a psychiatrist or primary care physician, while others allow self-referrals and conduct their own evaluation before treatment.
Insurance coverage for TMS typically requires documentation of prior treatment attempts, such as antidepressant trials or psychotherapy. Each insurance provider has specific medical necessity criteria that must be met before authorization is granted.
TMS must be prescribed and supervised by a licensed physician, usually a psychiatrist. Treatment is delivered in a clinical setting by trained staff under medical oversight. Regulations vary slightly by state, but TMS is not a home-based or direct-to-consumer treatment. It is considered a medical procedure requiring professional evaluation, documentation, and monitoring.
Putting TMS Into Context
TMS, or Transcranial Magnetic Stimulation, is built on a basic physical principle: a changing magnetic field can generate a small electrical current. When that magnetic field is delivered in a focused way to a specific part of the brain, it can influence the activity of the neurons in that region. Repeated over days and weeks, that stimulation may gradually alter the way those circuits function.
Within its FDA-cleared indications, TMS is a studied and regulated treatment supported by clinical trials and professional guidelines. It represents one approach among several in modern psychiatry. For individuals who have not found sufficient relief with medications, it offers a different method of engaging the brain’s mood-related networks.
If you are considering TMS, the next step is not assumption but evaluation. A qualified clinician can determine whether your diagnosis, treatment history, and medical profile make TMS a reasonable option within a broader care plan.
Understanding what TMS is, how it works, and where it fits allows you to approach it with clarity rather than hype. When expectations align with evidence, treatment decisions become more grounded and more informed.
References
- U.S. Food and Drug Administration (FDA).
510(k) Summary: NeuroStar TMS Therapy System for the treatment of Major Depressive Disorder. 2008. - U.S. Food and Drug Administration (FDA).
FDA clears TMS device for obsessive-compulsive disorder. 2018. - George MS, Lisanby SH, Avery D, et al.
Daily left prefrontal transcranial magnetic stimulation therapy for major depressive disorder: A sham-controlled randomized trial.
Archives of General Psychiatry. 2010;67(5):507–516. - O’Reardon JP, Solvason HB, Janicak PG, et al.
Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: A multisite randomized controlled trial.
Biological Psychiatry. 2007;62(11):1208–1216. - Gaynes BN, Lloyd SW, Lux L, et al.
Repetitive transcranial magnetic stimulation for treatment-resistant depression: A systematic review and meta-analysis.
Journal of Clinical Psychiatry. 2014;75(5):477–489. - Perera T, George MS, Grammer G, et al.
The Clinical TMS Society consensus review and treatment recommendations for TMS therapy for major depressive disorder.
Brain Stimulation. 2016;9(3):336–346. - Rossi S, Hallett M, Rossini PM, Pascual-Leone A.
Safety, ethical considerations, and application guidelines for transcranial magnetic stimulation in clinical practice and research.
Clinical Neurophysiology. 2009;120(12):2008–2039.